Geology is an interesting subject as it involves studying different features of the earth including the oceans, mountains, rocks, earthquakes, and volcanoes.
Basically, a geologist studies all solid and liquid matter that forms the earth in addition to the history and processes that have fashioned it. A basic and important part of geology is the earth’s interior. This part of geology is what we’re going to discuss here.
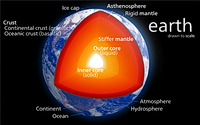
Earth’s Interior
Just like the interior of your car, the earth’s interior is made up of a variety of elements and a number of layers are present in the earth’s interior. The earth’s crust, mantle, and the core are its major layers. The mantle is further divided into the upper and lower mantle while a liquid outer core and a solid inner core constitute the earth’s core.
The earth’s crust
The first layer of the earth consisting of about 16 kilometers of rock as well as unattached materials is known as the crust. Compared to its thickness under the oceans, the crust’s thickness underneath the continents is almost three times more.
The Earth’s Mantle
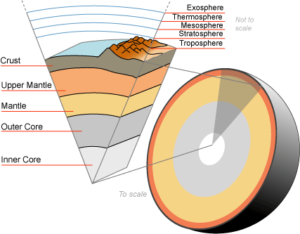 Mantle is the layer beneath the crust and accounts for a large majority of the Earth’s volume. Movements, know as convection in geology circles, is responsible for earthquakes and volcanic activity. Almost 1,800 miles deep, the mantle consists of substances that are rocky, solid and thick. Of the earth’s total weight and mass, 85 percent is made up of this substance. Very hard, rigid rocks make up the mantle’s first few miles. Super heated solid rock makes up the next 100 or so. Solid and sturdy rock materials make up the next hundred miles of the earth’s mantle.
Mantle is the layer beneath the crust and accounts for a large majority of the Earth’s volume. Movements, know as convection in geology circles, is responsible for earthquakes and volcanic activity. Almost 1,800 miles deep, the mantle consists of substances that are rocky, solid and thick. Of the earth’s total weight and mass, 85 percent is made up of this substance. Very hard, rigid rocks make up the mantle’s first few miles. Super heated solid rock makes up the next 100 or so. Solid and sturdy rock materials make up the next hundred miles of the earth’s mantle.
The Earth’s Core
Two layers that are mostly iron make up the earth’s core. Iron is estimated to make up about 90% of the earth’s core with oxygen, sulfur, or nickel combining to form the remaining 10 %. The earth’s core consists of mostly iron and nickel. Solid, the earth’s inner core measures about 1500 miles in diameter.
The earth’s outer core also consists of mostly nickel and iron. The earth’s inner and outer cores combine to become as big as the planet Mars. Superheated liquid molten lava is what most people believe earth’s outer core is made of. A solid ball comprising mainly of nickel and iron is believed to make up the earth’s inner core.
There you have it—the earth’s interior and its components. The next time you start digging holes in your garden, you will know exactly what lies underneath it.









